Infernal contradictions
How many beekeepers read the documentation that accompanies the miticides they use for Varroa control? How many understand what all the terms – including the pharmacological ones – mean?
Posology anyone?
What about the phrase “Withdrawal period”?
Can all miticides containing the same active ingredient be used in the same way? If not, why not?
What about repeat usage? Can you repeat a treatment (if needed) if the instructions do not explicitly state that repeat treatments are not allowed? {{1}} Or can you only administer a second application if the instructions explicitly state that it is allowed?
And if a you are allowed to apply a second treatment, can you administer a third? What about treating in November and the following January? Two different calendar years, but well under one year apart.
Don’t expect any answers to these or related questions in this post 😉 .
The intention here is to highlight the slightly shambolic nature of the documentation that accompanies (and sometimes does not accompany, but which you are probably expected to read!) the miticides approved for use in the UK. I don’t have time to cover all the miticides in a single post so will restrict this post {{2}} to two containing formic acid and one that contains oxalic acid.
And … while we’re at it … which are the legally binding instructions? Those in teensy-weensy lettering on the purchased product or the ones listed in the Veterinary Medicines Directorate (VMD) database?
MAQS and FormicPro
MAQS (Mite Away Quick Strips) and FormicPro are very similar products.
Actually, they are so similar that it’s rather difficult to tell them apart.
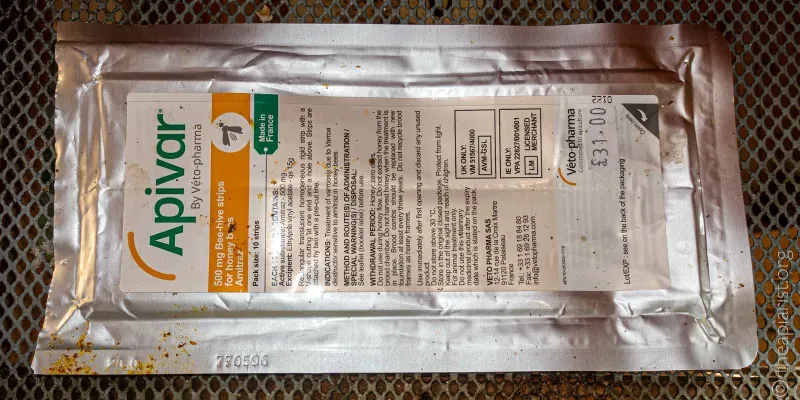
The packaging is similar – a cardboard box or plastic tub filled with sachets, each containing two strips impregnated with formic acid (and some other stuff – but what isn’t specified). Even the price is similar; two doses (by which they mean sufficient to treat two hives, or one hive twice, cost an eye-watering {{3}} £16.50. I’ve not checked other suppliers, but Thorne’s list the 2, 10 and 30 dose boxes of MAQS and FormicPro at identical price points {{4}}.
If you bother to read the online documentation (which you should) you will see that both are marketed by NOD Apiary Ireland Limited, and that each strip contains 68.2 g of formic acid. Even the description of the individual gel strips is very similar:
Brown, semi-rigid to soft gel strip covered in a biodegradable laminated paper, which maintains form (FormicPro).
Each strip is an off-white to caramel coloured gel wrapped in white laminated biodegradable paper (MAQS).
So, we have the same active ingredient, formulated in the same way, packaged in a similar manner, with identical diagrams for how to apply two strips to the brood box. The temperature range recommended for use is identical and both have similar warnings about queen damage.
The same but different
But, although MAQS and FormicPro appear to be essentially the same, from a practical beekeeping standpoint they are very different.
MAQS can be used with honey supers on the hive but FormicPro cannot.
Of course, pedantically, that’s not exactly true.
You could use them ’any-damned-way’ you please, but you would probably be breaking the rules.
You are allowed to use MAQS when there are honey supers present, but you are not allowed to use FormicPro – in all other regards an identical product – when there are honey supers on the hive.
Here are the relevant words from the SPC’s (Summary of Product Characteristics) {{5}};
Supers with honey must be removed from the hive prior to product application. See Section 4.5. Honey stored in super(s) put on for the treatment period must be removed and not used for human consumption. Spent strips must be removed before supers intended for harvest are placed on the hive (FormicPro – section 4.11 ‘Withdrawal period’).
The strips may be applied during honey flow; put on honey supers if honey flow is anticipated, to allow adequate space for colony expansion (MAQS – section 4.5 ‘Special precautions for use’).
There is one other difference as well … you can buy FormicPro whereas MAQS appears to be out of stock from all the suppliers I’ve checked.
Perhaps it has been withdrawn already by the manufacturer … ?
This is going to confuse a lot of beekeepers who have come to value MAQS as a short-term and effective treatment for Varroa management during the season.
Many will continue to use FormicPro in the same way that they used MAQS … which could be problematic if they are visited by a Seasonal Bee Inspector.
Summary of Product Characteristics (SPC)
Any miticides you purchase should be accompanied by a set of instructions – on the outside of the box, or the foil packet or wherever. These are often like ’ant tracks’ – illegibly small printing, almost impossible to read without the use of a binocular microscope {{6}}.
Importantly, the packet will also carry a lot number and a use by date – you need to keep records of the former for several years {{7}} after use. I almost always forget to write this into my notes, but I always photograph the packet so have a dated copy on my ‘phone.
If you want to review the official documentation for the miticide you need to visit the Veterinary Medicines Directorate Product Information Database. With a bit of rummaging around (Hint .. the search facility is your friend {{8}} ) you can usually find at least two official documents for each authorised product:
A document prefixed SPC (the Summary of Product Characteristics).
A document prefixed QRD (for Quality Review of Documents), which is the Product Literature; essentially the labelling and text that is supplied when you purchase the product.
If you read these you will find a large amount of duplication. These documents are periodically revised – the MAQS and FormicPro paperwork is all dated June 2022, with MAQS being first authorised in 2013 and FormicPro in 2021.
Discrepancies and confusion
Aside from the ‘biggy’ (not being allowed to use FormicPro when there are supers on the hive) there are other discrepancies or confusing text in these documents.
I’ve already listed one example …
The MAQS SPC indicates the ability to use the product when supers are present under section 4.5 ’Special precautions for use’.
In contrast, the FormicPro SPC indicates that the product cannot be used when honey supers are present under section 4.11 ’Withdrawal period’, though they do refer to section 4.5 (where, perplexingly, only empty honey supers are mentioned).
Section 4.5 seems to me to be the logical place to mention the ever-so-slightly-critical matter of not being allowed to use FormicPro when there are honey supers present.
Does anyone proof read or sanity check these documents?
If so, why don’t they ever define the term withdrawal period?
If you do a search online for ’withdrawal period’ there are all sorts of things about hormonal birth control and legal contract cancellations, but you need to scroll down to the penultimate item on the first page to get the relevant meaning:
The time that must elapse between the last administration of a veterinary medicine and the slaughter or production of food from that animal, to ensure that the food does not contain levels of the medicine that exceed the maximum residue limit.
And that’s from the European Medicines Agency; it wasn’t until somewhere on the third page of results I could find anything from the VMD {{9}}.
Helpful? Not 🙁 .
Of course, there’s an argument that if you’re applying the ‘medicine’ then you should understand all the paperwork and seek further advice if needed.
But I suspect many do not.
More vagueness
Whilst very specific in places e.g. duration of treatment, maximum temperature for use 29.5°C (Really? Does that 0.5°C make a difference? How many domestic thermometers are that accurate?), the documentation also carries other contradictory or vague instructions.
Both MAQS and FormicPro contain the following words under Section 4.4 (‘Special warnings for each target species ‘) of the SPC
Use according to local treatment recommendations, if available.
Who makes these local treatment recommendations? Are they legally binding? Can you just invent them? What can they cover or not cover? Could the local treatment recommendations state “Use five strips for a month”?
And what about disposal of the used, unused and waste products? Here you will find instructions in two separate places in the SPC.
When removed, dispose of by composting (FormicPro, Section 4.9 “Administration”).
The strips do not need to be removed from the hive after the application period of 7 days as the honey bees dispose of the spent strips. If they are removed, dispose of by composting (MAQS, Section 4.9 “Administration”).
And, confusingly …
Any unused veterinary medicinal product or waste materials derived from such veterinary medicinal products should be disposed of in accordance with local requirements (MAQS and FormicPro Section 6.6 ‘Special precautions for disposal’)
So can they be composted, or do ‘local requirements’ take precedence?
I can’t even be bothered to comment on section 4.6 ‘Adverse reactions’ which helpfully define very common, common, uncommon, rare etc events, but then only apply them to one adverse reaction, despite listing many others.
Grrrrr.
I’ve spent a career trying to make sense of badly worded, confusing, verbose, self-contradictory documents (until the arrival of ChatGPT this was the norm for both student essays and University administrative paperwork) but some of these instructions still baffle me.
Api-Bioxal
The active ingredient in Api-Bioxal is oxalic acid (OA). I’ve discussed this extensively in previous posts. There are several other miticides listed on the VMD database that have OA as the active ingredient; Oxuvar, VarroMed (which also contains formic acid), Dany’s BienenWohl powder/solution and Oxybee. Of these, the last two may not be routinely available in the UK.
I’m going to restrict my (brief) discussion to Api-Bioxal as it’s the only one I’m familiar with and because it highlights a different form of internal infernal contradiction in the official instructions and paperwork.
The Api-Bioxal SPC and instructions clearly state (in section 4.5 ’Special precautions for use’ … or ‘the logical place’ as it should be known) that it should be administered when supers are not present on the hive.
In addition, it also clearly states that the withdrawal period is ‘Zero days’ {{10}}.
The duration of application for MAQS and FormicPro is seven days and the formic acid permeates the cappings and kills mites in capped cells. In contrast, Api-Bioxal is a single shot treatment … it may (or may not) remain active in the hive for some time after administration, but you essentially apply it and walk away.
Job done 🙂 .
Oxalic acid does not penetrate capped cells and so is only effective if the colony is broodless. The instructions are clear on this point (to their credit).
A single shot used once … or twice?
The instructions describe two approved methods of administering Api-Bioxal. Trickling a 4.2% (w/v) solution made up in 1:1 (‘thin’) syrup onto the visible seams of bees, or vaporising a hive with up to 2.3 g of Api-Bioxal.
Administration by trickling … Up to two treatments per year (winter and/or spring-summer season in brood-free colonies). The treatment should be made in a single administration.
Administration by vaporisation … Maximal dose 2.3g per hive as a single administration. One treatment per year.
I think the ‘single administration’ means that you cannot split a treatment into two e.g. vaporise 1.15 g twice, or trickle 2.5 ml per seam and then repeat it the following day.
What’s odd is that trickling can be conducted twice per year, whereas vaporisation cannot. What about vaporising in December and January? i.e. once in each of two successive years … which could even be on successive days (31/12 and 1/1).
This is odd for two reasons – firstly it seems strange that the same compound can be administered a different number of times depending upon the route of administration.
Well, OK, perhaps it’s really bad for the colony to be vaporised? In that case it would be understandable, though some explanation of the point would help.
Trickling and vaporising do cause differential damage to colonies, but it is trickling that does more damage. Trickled OA damages open brood and studies from the LASI group in Sussex showed that colonies trickle-treated when brood was present were subsequently weaker than those that were vaporised (Al Toufailia et al., 2015).
Conversely, several studies of repeated vaporisation have shown that it is well tolerated by the colony.
So, in this instance the instructions are at odds with my understanding {{11}} of the current science.
Zero days
If the withdrawal period for Api-Bioxal is zero days (it is), can you add a stack of supers to the colony the day after vaporising or trickle treating a colony?
I think you can {{12}}.
Which is a little odd as the oxalic acid remains active in the colony for several days after it is added. If you apply Api-Bioxal and then monitor mite drop on a daily basis over about a week it often peaks a day or two after it is administered, but goes on at a reducing rate for ~5-6 days. Whilst it could just be taking its time killing the mites {{13}}, I think it is more likely that residual activity remains for several days.
Perhaps the wording in the instructions on ‘honey flow’ precludes this, but you can certainly add supers before a honey flow and I’d argue that the wording isn’t completely clear cut.
Licensing
I know almost nothing about the licensing of veterinary medicines. My understanding is that a license is applied for, supported by evidence of efficacy, toxicity etc. and that it is restricted in terms of the range of methods used to apply the miticide.
Therefore, if the manufacturer only applies for a license for trickling or vaporisation, then that’s what they get (if approved). Varromed (an OA solution) can be administered by trickling and spraying. When made up for spraying the OA solution has a long shelf life as there is no sugar present.
But that’s not an option for Api-Bioxal 🙁 .
Beekeepers are restricted in what they can (legally) do by what the manufacturer sought a licence for, even if there are better ways of administering the active compound, or even if the scientific evidence (sometimes preceding licensing, and certainly preceding updates of the documentation) indicates that – for example – repeat administration is both safe and effective.
Trying to make sense of it all
In Scotland a Working Group has been established to try and resolve some of these discrepancies and provide better advice to beekeepers on the use of the currently licensed miticides.
The Working Group involves representatives from a variety of interested parties including an acronym salad comprising SASA, VMD, BFA, FSS, SBA, SRUC, SEPA, APHA, DEFRA, DAERA, NBU and some academics and ex-academics with a particular interest in honey bee health.
I have written a lot about Varroa control on this site. In my view it is relatively straightforward to control mite numbers using the currently licensed miticides appropriately. In my experience it is easier to do this in Scotland, where we have lower winter temperatures and a greater chance of an extended broodless period.
However, Scotland – unlike the Midlands where I have also kept bees – offers some additional complications where Varroa control is concerned. Our most important (by £££) nectar source is heather which yields late in the year, too late in some years to subsequently protect the winter bees from mites and viruses.
Balancing the needs of the bees (low mites and viruses to overwinter successfully) with those of the beekeeper (hundreds of kilograms of heather honey) requires a careful balancing act and a good understanding of the benefits and limitations of the miticides available.
In turn, this needs good documentation and better advice that is both easily accessible and understandable by beekeepers.
And … to my surprise – and I look forward to it being confirmed or refuted – I’m told that the SPC is the legally binding document with regard to the use or misuse of licensed miticides.
I’ve (had to) read them all now … have you?
References
Al Toufailia, H., Scandian, L., and Ratnieks, F.L.W. (2015) Towards integrated control of varroa: 2) comparing application methods and doses of oxalic acid on the mortality of phoretic Varroa destructor mites and their honey bee hosts. Journal of Apicultural Research 54: 108–120 https://doi.org/10.1080/00218839.2015.1106777.
{{1}}: Too many double negatives there, sorry.
{{2}}: The first in a series of 41.
{{3}}: That’s a pun based upon the chemical content … whilst expensive for a small amount of formic acid, this is insignificant when compared with the financial hit if you lose the colony.
{{4}}: £58 and £155 for 10 and 30 doses respectively, a compelling reason to buy with fellow beekeepers if you only have a few hives.
{{5}}: More on these documents shortly.
{{6}}: Or perhaps I just need even stronger glasses?
{{7}}: Five? I can’t remember off the top of my head.
{{8}}: Limit your search to ‘species = bees’ to avoid the horse tranquillisers, dog worming tablets and treatments for scaly face in budgerigars.
{{9}}: And that was a withdrawn document.
{{10}}: Of course, there’s no explanation what this means.
{{11}}: And I’m not the only one.
{{12}}: And, as it’s zero, what about immediately?
{{13}}: Serves them right!






Join the discussion ...