Repeated oxalic acid vaporisation

Repeated oxalic acid vaporisation: methods that work, and why they work
Oxalic acid is a simple chemical. A dicarboxylic acid that forms a white crystalline solid which dissolves readily in water to form a colourless solution. It was originally extracted from wood-sorrels, plants of the genus Oxalis, hence the name. In addition to the wood-sorrels it is present in a wide range of other plants including rhubarb leaves (0.5% oxalic acid {{1}} ), the berries and sap of Virginia creeper and some fruits, such as starfruit. Additionally, fungi excrete oxalic acid to increase the availability of soil nutrients.
Oxalic acid is inexpensive to produce by a variety of processes and was possibly the first synthesised natural product. About 120,000 tonnes are produced annually and it is mainly used for bleaching wood (and often sold as ‘wood bleach’) and cleaning products – including teeth. It chelates iron and so is used for rust removal and is used as a dye fixative (or mordant {{2}} ).

Oxalic acid and API-Bioxal … the same but different
It is also, when used properly, devastatingly effective against the ectoparasitic mite Varroa destructor.
And, even more importantly, when used properly it is extremely well-tolerated by honey bees.
Great!
Not so fast …
Unfortunately for beekeepers, some of the commercially available i.e. licensed and approved, oxalic acid-containing treatments either contain unnecessary additives and/or have limitations in their approved modes of administration that reduces their efficiency and use in real world beekeeping situations.
Oxalic acid-containing miticides and their use
A quick search of the UK’s {{3}} Veterinary Medicines Directorate snappily titled Product Information Database for ‘target species = bees’ and ‘active ingredient = oxalic acid’ yields three products :
- Varromed (BeeVital GmbH) which is a solution containing formic acid and oxalic acid
- Oxybee (DANY Bienenwohl GmbH) which is an oxalic acid solution PLUS a separate powder containing essential oils and sugar. As far as I can tell, Oxybee looks to be the same product as Dany’s BienenWohl powder and solution, which – although listed and licensed – I cannot find for sale {{4}} in the UK
- API-Bioxal (Chemicals Laif S.P.A) which is purchased as a powder composed of 88% oxalic acid dihydrate together with silica and glucose
I’m going to largely ignore Varromed and Oxybee for the rest of this post. I’m sure they’re perfectly good products but I’ve not used either of them so cannot comment from personal experience.
Keeping your powder dry
More relevant to this post, Oxybee and Varromed are both liquids, and this post is about vaporising (aka sublimating) oxalic acid.
And vaporisation involves using the powdered form of oxalic acid.
Which neatly brings me to the methods of application of oxalic acid-containing treatments to kill mites.
I’m sure there are some weird and wonderful ones, but I’ll be limiting any comments to just three which – from my reading of the instructions – are the only ones approved (and then not for all of the products listed above) : {{5}}
- Spraying a solution onto the surface of the bee-covered frames
- Dribbling or trickling a solution onto each seam of bees between the frames
- Vaporisation or sublimation of powdered oxalic acid by heating it in a metal pan to convert it to a gas. This permeates the hive, settling on all the surfaces – woodwork, comb, bees – and remains active against mites for a period after administration
Broodless is best
Oxalic acid, however it is administered, does not penetrate brood cappings. Therefore all of the approved products are recommended for use when the colony is broodless.
Typically – though not exclusively – this happens in the winter, but the beekeeper can engineer it at other times of the season.
If the colony is broodless you can expect any oxalic acid-containing miticide to reduce the mite population by 90% or more. There are numerous studies that support this level of efficacy and it’s what you should be aiming for to give the colony the best start to the season.
I discussed at length how to determine whether a winter colony is broodless a fortnight ago in Broodless?
This post is a more extensive response to several comments (made to that Broodless? article) that recommended repeated vaporisation of oxalic acid at, either 4, 5 or 7 day intervals.
The idea is that this kills the phoretic mites present when the colony is first treated and the mites subsequently released as brood emerges.
How many repeats?
I’ve seen anything from two to seven recommended online.
I’ll discuss this further below, but I’d note that the very fact that there’s such variation in the recommended repeat treatments – perhaps anything from two, fours days apart to seven at weekly intervals (i.e. spanning anything from 8 days to 49 days) – suggests to me that we don’t know the optimal treatment schedule.
Which is a little weird as, a) Varroa is a globally-distributed problem for beekeepers and is more or less invariant (as is the brood cycle of the host honey bee), and b) repeated treatment regimes have been used for over 20 years.
Which brings me back to a crude comparison of vaporisation vs dribbling, or …
Sublimation vs. trickling
A hive can be sublimated with oxalic acid without opening the hive. The vaporiser alone is introduced through the hive entrance or – in the case of certain models – the vapour is squirted through a hole in the floor, brood box or eke. In contrast, trickling oxalic acid requires the removal of the crownboard.
In the video above I’m using a Sublimox vaporiser. The hive entrance is sealed with foam and the open mesh floor is covered with a tightly fitting slide-in tray. As you can see, very little vapour escapes.
Although oxalic acid is well tolerated by bees, and it has no effect upon sealed brood, a solution of oxalic acid is detrimental to open brood. Therefore, trickled oxalic acid weakens the colony – because the acidity kills some or all of the open brood – and repeated trickling of oxalic acid is likely to compound this (see Al Toufailia et al., 2015). In contrast, repeated oxalic acid vaporisations appear not to be detrimental to the colony (caveat … I’m not aware of any long-term studies of this, or for the impact on the queen).
API-Bioxal approved methods of administration
The instructions for API-Bioxal clearly state that only a single treatment by vaporisation is approved per year. The exact wording is:
Maximal dose 2.3g per hive as a single administration. One treatment per year.
In contrast, when used as a solution for trickling the instructions state:
Up to two treatments per year (winter and/or spring-summer season in brood-free colonies).
This seems nonsensical to me considering what we now know about oxalic acid – remember, API-Bioxal was licensed in the same year (2015) that Al Toufailia et al., demonstrated it was detrimental to open brood, and I’m reasonably sure this had been shown previously (but can’t currently find the reference).
But, it gets worse …
API-Bioxal contains oxalic acid with powdered silica and glucose. I presume the silica is to keep it free-running. I’m not aware that powdered silica kills mites and I’m damned certain that glucose has no miticidal activity 😉 .
Neither of these two additives – which I’ve previously called cutting agents – are there to increase the activity of the oxalic acid … and the presence of the glucose is a real problem when vaporising.

Caramel coated Sublimox vaporiser pan
When glucose is heated to 160°-230°C it caramelises (actually, this happens at 150°C {{6}} ), coating the inside of the vaporising pan. This needs to be cleaned out afterwards {{7}}. The instructions state:
Cool down and clean the vaporizer after use to remove possible residue (max 6%, around 0.140 g).
However, I don’t want to focus on what I consider to be a very effective but decidedly sub-optimal product … instead I want to discuss whether repeat treatment with oxalic acid actually works when there is brood present.
Why is repeat treatment recommended?
Remember, it’s not recommended or approved by the manufacturers of API-Bioxal or the Veterinary Medicines Directorate. I really should have titled this section ’Why is repeat treatment recommended by those who advocate it?’
But that wouldn’t fit on a single line 😉 .
When you sublimate oxalic acid, the gas cools and the oxalic acid crystals settle out on every surface within the hive – the walls, the frames, the comb, the bees etc.. For this reason, I prefer to vaporise oxalic acid when the colony is not tightly clustered. I want everything to be coated with oxalic acid, and I particularly want every bee to be coated because that’s where most of the mites are.
Unless they’re in capped cells 🙁 .
And if they’re in capped cells, the only way the Varroa (released when the brood emerges) will come into contact with oxalic acid is if it remains present and active within the hive. Unfortunately, it’s unclear to me exactly how long the oxalic acid does remain active, or what accounts for a drop in its activity.
But it does drop.
If you treat a colony with brood present and count the mites that appear on the Varroa tray every day it looks something like this:
Mite drop per day before and after treatment
’Something like’ because it depends upon the phoretic mite levels and the amount and rate of brood uncapping. For example, you often see higher mite drops from 24-48 hours than 0-24 hours after treatment.
I know not why.
The drop in the first 48 hours – presumably almost all phoretic mites – can be very much higher than the drop from day three onwards {{8}}.
The duration of activity after vaporisation
Some studies claim oxalic acid remains active for 2-3 weeks after administration. I’m a little sceptical that it’s effective for that long and my own rather crude observations of post-treatment mite drop (of brooding colonies) suggests it returns to background levels within 5-7 days.
I could rabbit on about this for paragraphs as I’ve given it a reasonable amount of thought, but fortunately the late Pete Little did the experiment and showed that:
The recommended dose for colonies with brood is three or four doses seven days apart, however I found out that this is not effective enough, and treated 7, 6, 5 4, 3, 2 days apart to find out the most effective which is 5.
It therefore makes sense that three treatments at five day intervals should be sufficient. This period comfortably covers a complete capped brood cycle (assuming there is no drone brood in the colony) which is 12 days long.
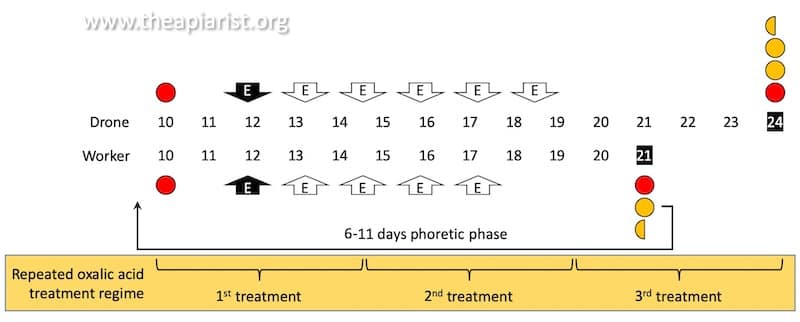
Repeated oxalic acid vaporisation treatment regime.
If there is drone brood present you would theoretically need four treatments at 5 day intervals to be sure of covering the 15 day capped brood cycle of drones.
But it turns out there are some additional complications to consider.
Dosage
In the UK the recommended i.e. approved, maximum dose of API-Bioxal is 2.3 g by vaporisation. Remember my comments about the other rubbish stuff API-Bioxal contains, 2.3 g of API-Bioxal actually contains a fraction over 2 g of oxalic acid dihydrate.
This is the active ingredient.
When comparing different experiments where some have used ‘plain’ oxalic acid dihydrate and others have used – or will use – API-Bioxal, it’s important to consider the amount of the active ingredient only {{9}} .
In the US, oxalic acid was registered as an approved treatment for Varroa in 2015. By vaporisation, the approved dosage is 1 g of oxalic acid dihydrate per brood box i.e. half that approved in the UK.
Remember also that a deep Langstroth is 5% larger (by volume) than a National brood box.
And Jennifer Berry and colleagues in the University of Georgia have recently determined whether repeated administration of vaporised oxalic acid to a colony rearing brood is an effective way of controlling and reducing Varroa infestations (Berry et al., 2021).
And the answer is … decidedly underwhelming
Here are the experimental details.
The paper doesn’t state {{10}} when the experiment was done but they measured honey production in the treated colonies and were definitely brood rearing, so I’m assuming late summer.
Colonies were treated with 1 g / box (double Langstroth deeps) vaporised oxalic acid every five days for a total of 35 days i.e. 7 applications. Mite infestation levels (percent of workers carrying phoretic mites) were measured before and after treatment. Almost 100 colonies were used in the experiment, in three apiaries, randomly split into treated and control groups.
Let’s get the easy bit out of the way first … there was no difference in brood levels, adult bees or food stores at the end of the study. The treated hives were not disadvantaged by being treated … but they didn’t gain an advantage either 🙁 .
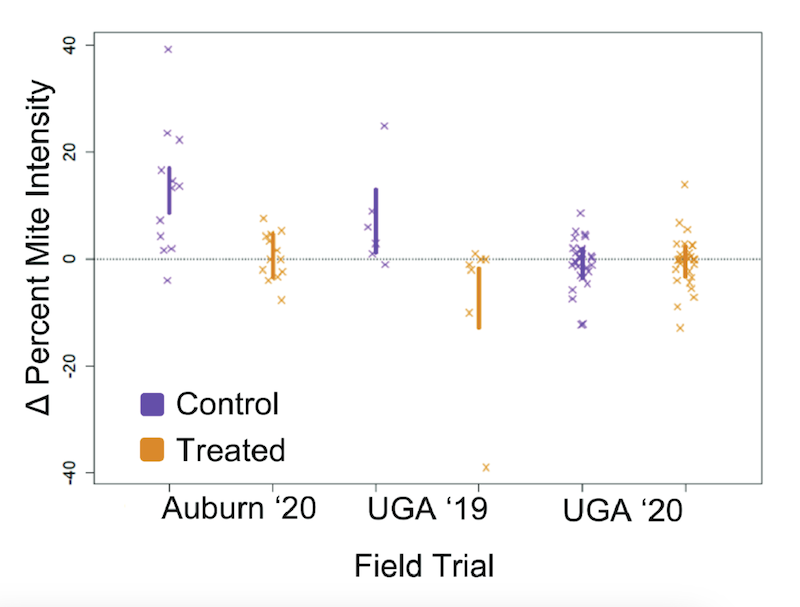
Mite levels after treatment normalised to pre-treatment levels (dotted line = no change)
During the experiment the percent mite infestation (PMI) levels in the untreated control colonies increased (as expected) by ~4.4. This is an average and there was quite a bit of variation, but it means that an initial mite infestation level of 4 (average) increased to 8.4 i.e. over 8 mites on every 100 adult workers in the hive.
3% is often considered the cutoff above which treatment is necessary.
Overall, the PMI of treated colonies reduced over the duration of the experiment … but only by 0.7.
From a colony health perspective this is a meaningless reduction.
Seven treatments with the recommended (in the US) dose of oxalic acid stopped the mite levels increasing, but did not reduce them.
Repeated administration of the US-approved oxalic acid dose by vaporisation does not reduce mite levels in a way that seems likely to significantly benefit the colony.
🙁
Dosage, again
I’m not sure the primary data used to justify the US approved 1 g / box dosage. Early studies by Thomas Radetzki (PDF) showed a 95% reduction in mite levels using a dose of 1.4 g. This was a large study involving ~1500 colonies and a dose of 2.8 g was not significantly more effective. I’m quoting the figures for broodless colonies {{11}}.
The Berry results were similar to two smaller previous studies by Jamie Ellis and colleagues (Jack et al., 2020, 2021) who demonstrated that 1 g oxalic acid vaporised three times at weekly intervals was ineffective in controlling mite levels.
However Jack et al., (2021) also applied a similar treatment schedule using different doses of oxalic acid.
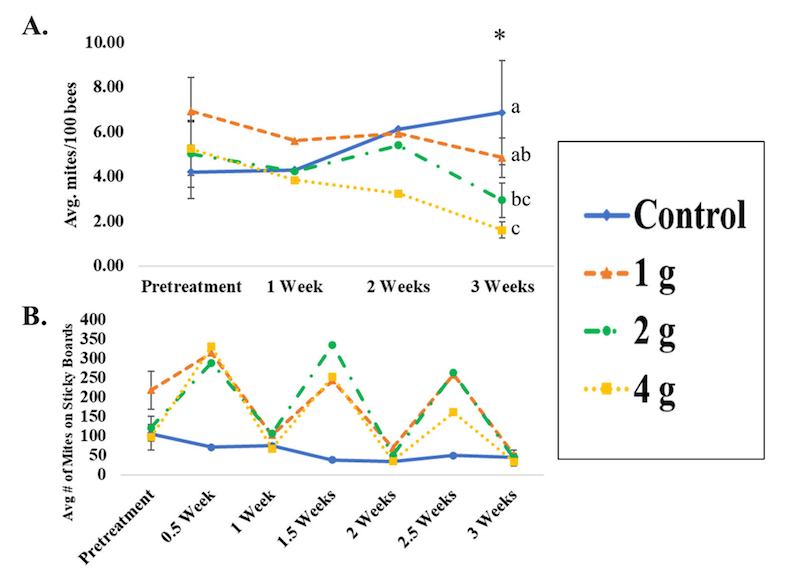
Data from Jack et al., 2021 using different repeat doses of oxalic acid
Ignore the intermediate values in panel A, just look at the pretreatment and ‘3 weeks’ mite infestation values.
Mite levels increased in untreated controls and decreased in all treated colonies. However, there was a clear dose response where the more oxalic acid used the greater the impact on the mite levels.
Four grams of oxalic acid reduced the mite infestation rate significantly … from ~5% to ~2% (I’ll return to this). However, the intermediate levels of oxalic acid, whilst reducing mite levels, did not do so significantly from the next closest amount of oxalic acid. For example, 1 g wasn’t significantly more effective than no treatment (as already stated), 2 g was not significantly more effective than 1 g and 4 g was not significantly more effective than 2 g.
But wait … there’s more
I’m familiar with two other studies that look at dose and/or repetition and efficacy (there are more, but this isn’t meant to be an exhaustive review, more a ”Do we know enough?” overview).
Gregoric et al., (2016) published a {{12}} study that appeared to use combinations of treatments in multiple apiaries. The abstract claims 97% reduction using three 1 g vaporisations, though these are spread over a 57 day period (!) stretching from mid-August to late-November. Mite drop in November following treatment was ~75% (presumably broodless) , but only 10-20% in August. Interestingly I can’t find the figure 97% anywhere in the results …
Finally, Al Toufailia et al., (2015) investigated the dose response to vaporised oxalic acid, showing an 80% reduction in infestation at 0.56 g and 93-98% who using 1.125, 2.25 and 4 g of oxalic acid. All of these studies were determined using broodless colonies.
The Al Toufailia and Jack studies – as well as the Berry study – also reported on adverse effects on the colony. With certain exceptions vaporisation was well tolerated. Some colonies went queenless. Where the queen was caged in late summer to render it broodless (Jack et al.,) some colonies subsequently failed to overwinter successfully (though, look on the bright side, mite levels were reduced 😉 ).
Don’t do that at home … I presume they impacted the production of winter bees.
confused.com
I’m not sure there’s a compelling, peer-reviewed study that definitively shows that repeat treatments of vaporised oxalic acid administered to a brood rearing colony reduces mite levels sufficiently.
Yes, the Jack et al., (2020) showed a significant reduction in the infestation rate (using 4 g three times at seven day intervals), but it was still around 2%.
In late summer, with 20-30,000 bees in the box and 6 frames of brood, that’s still ~600 mites (and potentially more in the capped brood).
In midwinter with about 10,000 workers and much smaller amounts of brood in the hive a 2% infestation rate is still 200 mites.
That’s still a lot of mites for a nearly broodless colony … I treat my colonies when broodless (and assume I’m killing ~90% of the mites present) and am disappointed if there are 45 mites on the Varroa tray. 50 mites on 10,000 workers is an infestation rate of 0.5%.
I’ve waffled on for too long.
All those advocating – or using – repeated oxalic acid vaporisation on brood rearing colonies in late autumn/winter need to think about:
- dosage … 1 g is clearly too little (at a 5-7 day interval, but what if it was at a 4 day interval?), 2 g is better and 4 g is well-tolerated and certainly more effective
- frequency … which I suspect is related to dosage. The goal must be to repeat sufficiently frequently that there is never a period when oxalic acid levels fall below a certain amount (and I don’t know what that amount is). 1 g on a daily basis might work well … who knows?
- duration … you must cover a full capped brood cycle with the repeats
- adverse effects … inevitable, but can be minimised with a rational treatment schedule
Broodless is best
It really is.
But, if your colonies are never broodless {{13}} then I wouldn’t be confident that repeat treatment was controlling Varroa levels sufficiently.
I have treated repeatedly with oxalic acid. In the good old days before API-Bioxal appeared. It certainly reduced Varroa levels, but not as well as my chosen Apivar does these days.
Repeated oxalic acid vaporisation is regularly proposed as the solution to Varroa but I’m certainly not confident that the data is there to support this claim.
Take care out there 😉
Notes
In a future post I’ll revisit this … I’ve got a pretty clear idea of how I’d go about demonstrating whether repeated oxalic acid treatments are effective in meaningfully reducing mite levels i.e. sufficient to protect the colony overwinter and through to the following late summer.
References
Al Toufailia, H., Scandian, L. and Ratnieks, F.L.W. (2015) ‘Towards integrated control of varroa: 2) comparing application methods and doses of oxalic acid on the mortality of phoretic Varroa destructor mites and their honey bee hosts’, Journal of Apicultural Research, 54(2), pp. 108–120. Available at: https://doi.org/10.1080/00218839.2015.1106777.
Berry, J.A. et al. (2022) ‘Assessing Repeated Oxalic Acid Vaporization in Honey Bee (Hymenoptera: Apidae) Colonies for Control of the Ectoparasitic Mite Varroa destructor’, Journal of Insect Science, 22(1), p. 15. Available at: https://doi.org/10.1093/jisesa/ieab089.
Gregorc, A. et al. (2016) ‘Integrated varroa control in honey bee (Apis mellifera carnica) colonies with or without brood’, Journal of Apicultural Research, 55(3), pp. 253–258. Available at: https://doi.org/10.1080/00218839.2016.1222700.
Jack, C.J., van Santen, E. and Ellis, J.D. (2020) ‘Evaluating the Efficacy of Oxalic Acid Vaporization and Brood Interruption in Controlling the Honey Bee Pest Varroa destructor (Acari: Varroidae)’, Journal of Economic Entomology, 113(2), pp. 582–588. Available at: https://doi.org/10.1093/jee/toz358.
Jack, C.J., van Santen, E. and Ellis, J.D. (2021) ‘Determining the dose of oxalic acid applied via vaporization needed for the control of the honey bee (Apis mellifera) pest Varroa destructor’, Journal of Apicultural Research, 60(3), pp. 414–420. Available at: https://doi.org/10.1080/00218839.2021.1877447.
{{1}}: No, I don’t think rhubarb leaves under the crownboard will do your colony much good – or harm for that matter – but if you really want to try then use parsley instead, as this contains over 1.7% oxalic acid.
{{2}}: From the Latin mordere ‘to bite’ … reflecting the attachment of the dye to the fabric.
{{3}}: Sorry readers, I can’t do this for other countries.
{{4}}: After a rather cursory search admittedly.
{{5}}: Confused, you soon will be.
{{6}}: Just to keep the pedants happy.
{{7}}: Or create a tightly-fitting tinfoil lining before use, which you can simply discard afterwards.
{{8}}: In case you can’t tell, the graph above is a cartoon – not real data.
{{9}}: As an aside, I think the API-Bioxal instructions are incorrect – I calculate that, using the maximum dose (which you should – see below) – there are 0.26 g of crap to be scrubbed out of the vaporiser.
{{10}}: Or my eyes are too tired to determine.
{{11}}: Where colonies were known to be rearing brood the figures were surprisingly (inexplicably and with no attempt made to justify them that I can see) high … let’s move on.
{{12}}: To me almost incomprehensible
{{13}}: Which is not the same as not broodless … if there is a period when they are broodless then ACT. Don’t decide to treat on a particular date if there’s a chance they will be broodless on a later or earlier date.

Join the discussion ...