A virus that changes bee behaviour
Particle physicists might not agree, but I think that evolution is the most powerful force in the universe. It is responsible for the fabulous diversity of life, for everything from the 6,000,000 kg Pando clonal colony of quaking aspen covering 43 hectares of the Fishlake National Forest in Utah, to the teeniest of tiniest of viruses.
As a microbiologist I’m acutely aware of the role evolution has played in the genetic arms race between hosts and pathogens. This is what is responsible for the multi-faceted immune system higher organisms carry – the antibodies, the lymphocytes, the complement and interferon responses, and everything else.
In turn, the fast replicating bacteria and viruses have evolved countermeasures to subvert these immune mechanisms, to switch them off entirely or to decoy them into targeting the wrong thing. This ‘arms race’ has gone on since well before the evolution of multicellular organisms (~600 million years ago) … and continues unabated.
Evolution is powerful for one simple reason; if a particular genetic combination {{1}} ‘works’ it will be passed on to the progeny. If a virus evolves a way to resist the immune response of the host, or to spread between hosts more efficiently, then the trait will be inherited.
Molecular mechanisms and behavioural changes
Some of changes work at the molecular level, invisible without exquisitely sensitive in vitro analysis; protein A binds to protein B and, in doing so, stops protein B from doing whatever it should have be doing. These are important but often very subtle.
Other changes are far more obvious. Take rabies for example (or don’t, it’s not recommended as it has a near-100% case fatality rate) … the primary host of the rabies virus are carnivorous mammals. Infection causes gross behavioural changes that facilitate virus transmission. The animal becomes bolder and much more aggressive, resulting in virus transmission through biting.
Recent studies have elegantly demonstrated that this (also) is an example of protein A binding to protein B at the molecular level, it’s just that the phenotype {{2}} is very much more marked.
A protein on the surface of the virus resembles a snake venom toxin and has the ability to bind to nicotinic acetylcholine receptors present in the central nervous system of the mammalian host. These receptors ‘do what they say on the tin’ and bind the neurotransmitter acetylcholine. If the virus protein binds the receptor the response to acetylcholine is blunted and this, in turn, leads to hyperactivity, one of the key behavioural responses caused by rabies viruses.
That’s enough about mad dogs.
If virus-induced behavioural changes are so obvious, why haven’t lots of different examples already been identified and characterised?
Sniffles
Part of the problem is the blurring of distinctions between overt behavioural changes and the direct symptoms induced due to the virus replicating.
Human rhinovirus, the aetiological agent of the common cold, causes upper respiratory tract infections. You get a runny nose and you sneeze a lot.
Your behaviour changes.
However, it’s generally accepted that the sneezing and runny nose are a result of the physiological response to infection, rather than a virus-induced behavioural response to facilitate transmission.
It’s worth noting that all that “stuff” that comes out of your nose contains infectious virus, so it’s perhaps an artificial distinction between the general symptoms of sickness and evolutionarily-selected host behavioural changes caused by the virus.
Which in a roundabout way …
… allows me to finally introduce the topic of bee viruses that cause host behavioural changes involved in their transmission.
Or, rather, one bee virus that does this … though I’m certain that there will be more.
Israeli Acute Paralysis Virus (IAPV) is an RNA virus transmitted horizontally by direct contact between bees, or while feeding on developing pupae, by the parasitic mite Varroa destructor. It was implicated as a causative agent of Colony Collapse Disorder (CCD), though really compelling evidence supporting it as the primary cause never materialised.
It’s a virus UK beekeepers should be aware of, but unworried by, as it is extremely rare in the UK.
A recent paper has shown two behavioural changes in response to IAPV infection in honey bees. One of them – that facilitates horizontal transmission between colonies – is also partially explained at the molecular level.
The paper was published a couple of months ago:
Geffre et al., (2020) Honey bee virus causes context-dependent changes in host social behavior. Proc. Natl. Acad. Sci. USA 117:10406-10413. {{3}}
I’m going to focus on the results, rather than the methods, though the methods are rather cool. They used barcoded bees to allow the automated image analysis of every bee in a colony for some of the studies where they had introduced known IAPV infected individuals.
Responses of nestmates to IAPV infected bees
Imagine watching a few hundred waggle dances and being able to recount the position, distance and response of every bee ‘watching’ {{4}} the dance, and then being able to summarise the results.
Over five days.
Non-stop.
Including nights (and yes, bees do still waggle dance at night – a subject for the future).
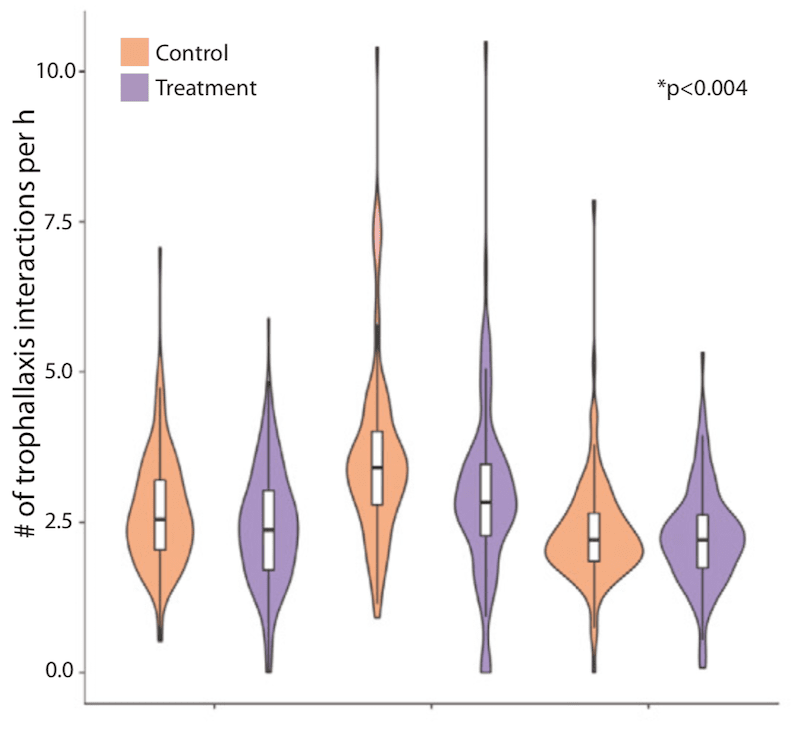
The scientists orally infected groups of 30 bees with a sub-lethal dose of IAPV, marked them and released them into an observation hive. They then recorded their movements around the hive and their interactions with other bees in the colony. In particular, they focussed on trophallaxis interactions where one bee ‘feeds’ another.
Trophallaxis is also considered to be a method of communication in the hive and has been implicated in disease transmission.
The authors love their whisker plots and statistical analysis.
Who doesn’t? 😉
However, they generally make for rather underwhelming images in a bee blog for entertaining reading. Here .. see what I mean …
Suffice to say that the results obtained were statistically significant.
They showed that the infected bees in the colony actually moved about the colony more than their nestmates. Conversely, they were engaged in fewer trophallaxis interactions i.e. it appeared as though they were being ‘ignored’ by their nestmates.
Were they really being ignored altogether or did their nestmates approach them, detect something was amiss and move away?
Antennation
Antennation is the mechanism by which bees recognise nestmates. They use the sensitive chemoreceptors on their antenna to detect cuticular hydrocarbons (CHC) which are distinctive between bees from different hives.
Antennation is a precursor to trophallaxis.
After all, bees do not want to feed a foreigner, or exchange chemicals involved in communications, or even potentially risk being exposed to a new pathogen.
Good as the barcoding and camera system is, it’s not good enough to record antennation within the observation hive. To do this they manually {{5}} recorded antennation events between IAPV-infected bees and nestmates in cages in the laboratory {{6}}.
In these studies IAPV-infected bees were engaged in the same number of antennation events as control bees. This strongly suggests that the nestmate could detect there was something ‘wrong’ with the IAPV-infected individuals. In support of this conclusion, the authors also demonstrated that bees inoculated with a double stranded RNA (dsRNA) stimulator of the honey bee immune response were also also antennated equally, but engaged in less trophallaxis interactions.
Therefore, these studies appear to show that nestmates exhibit a behavioural response to IAPV-infected bees (and bees with elevated immune responses, recapitulating their response to pathogen infection) that is likely to be protective, reducing the transmission of horizontally acquired viruses.
It’s worth noting two things here.
- There were no virus transmission studies conducted. It’s assumed that the lack of trophallaxis reduces virus transmission. That still needs to be demonstrated.
- This response is not induced by the virus on the host. It’s a response by nestmates of the host to virus infected individuals (or individuals that present as ‘sick’). As such it’s not the same as the rabies example I started this post with.
Virus-induced behavioural responses
But do the IAPV-infected bees behave differently when they come into contact with other bees who are not their nestmates?
After all, IAPV is a pathogenic virus and its continuing presence within a population (not just a single hive) depends upon it being spread from hive to hive.
For a highly pathogenic virus this is very important. If you spread from bee to bee within a hive and kill the lot you also go extinct … this partly explains the mechanism by which highly virulent viruses become less virulent over time.
But back to IAPV. What happens when IAPV-inoculated bees interact with bees from a different hive?
For example, what would happen if they drifted from one hive to another in a densely populated apiary? Drifting is a significant contributor to the spread of bees between adjacent colonies – studies show that 1% of marked bees drift to adjacent hives over a 3 day window. This partially accounts for the genetic mix of workers (up to 40% are unrelated to the queen that heads the colony) in a hive, a fact generally unappreciated by beekeepers.
The authors first showed that IAPV-infected bees could apparently leave and return to the hive with a similar frequency as uninfected foragers. Their flying was not compromised.
They then resorted again to recording interactions in the laboratory between IAPV-infected bees or control dsRNA-inoculated bees and workers from a different hive.
This was where it gets particularly interesting.
Hello stranger
The dsRNA-immunostimulated bees (remember, these induce a generalised immune response characteristic of a ‘sick’ bee) were treated aggressively by unmatched workers from a different hive.
In contrast, the IAPV-infected bees (which were ‘sick’ and would have been undergoing immunestimulation caused by the IAPV infection) experienced significantly less aggression than both uninoculated workers (which induced an intermediate response) and the dsRNA-inoculated.
This strongly suggests that IAPV is somehow able to modulate the appearance or behaviour (and one often determines the other) of the host to make it more acceptable to an unmatched worker.
They extended this study to conduct “field-based assays at the entrances of three normally managed honey bee colonies”, monitoring whether IAPV-infected bees were more likely to be accepted by the guard bees at the entrance of the hive.
They were. The IAPV-infected bees received a less aggressive reception and/or entered the hives much more easily than the controls.
But what about proteins A and B?
Good question.
The behavioural alterations described above must be explainable in terms of the molecular changes that IAPV induces in the bees. By that I mean that the virus must make, or induce the making of, a chemical or protein or other molecule, the presence of which explains their acceptance by the foreign guard bees.
And the obvious candidates are the cuticular hydrocarbons (CHC) that are recognized during antennation, which I introduced earlier.
And here the story leaves us with some tantalising clues, but no definitive answer.
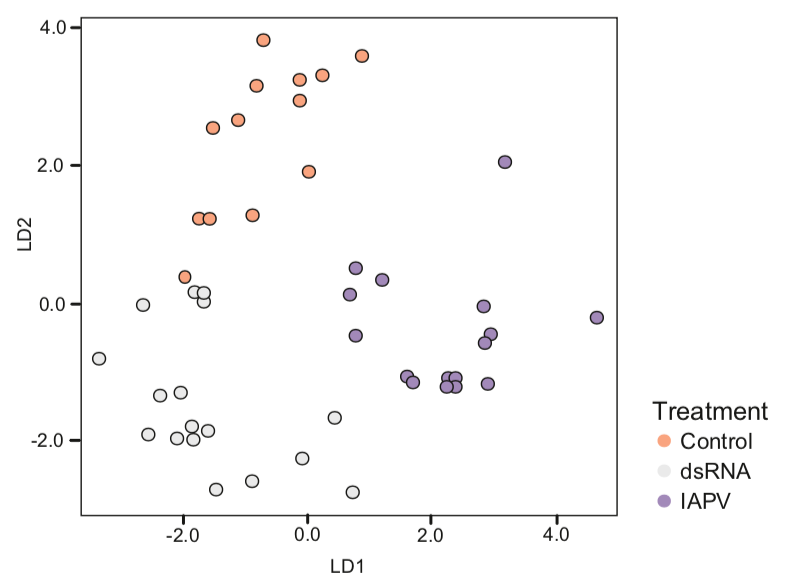
The scientists demonstrate that there were marked differences between the CHC profile of IAPV-infected and control bees. Again, they used their favoured whisker plots to show this, but collated all of the CHC data into an even more difficult to explain scatter plot of linear discriminant analysis.
The key take home message here is that for each of the CHC’s analysed there were differences in both the quality and relative abundance between the control bees, bees immunestimulated with dsRNA and the IAPV-infected bees.
These differences were so marked that you can see distinct clustering of points in the analysis above … these bees ‘look’ {{7}} different to the guard bees that antennate them.
This is a great story.
It’s as yet incomplete. To complete the understanding we will need to know which of those CHC’s, or which combination, when suppressed (or overrepresented) induce the guard bees to say “Welcome, step this way … “.
We’ll then of course need to find out how IAPV induces the change in CHC profile, which takes us right back to protein A and protein B again.
Ever the pedant
Much as I like this science I’d perhap argue that, again, the virus isn’t directly inducing a behavioural change in the host.
What it’s doing is inducing a behavioural change in the response to the infected host (by the guard bee). So perhaps this again isn’t quite the same as the rabies example we kicked off with.
A behavioural change in the host might include IAPV-infected bees drifting more, or drifting further. Alternatively, perhaps a colony with widespread IAPV infection could more easily indulge in robbing neighbouring colonies as they would experience less aggression from guard bees.
I can see immediate evolutionary benefits to a virus that induced these types of behavioural changes. It’s not an original idea … the late Ingemar Fries suggested it in a paper two decades ago {{8}}.
I’m also certain that researchers are looking for evidence supporting these types of directly-induced behavioural changes caused by viral pathogens in honey bees.
All religion, my friend, is simply evolved out of fraud, fear, greed, imagination, and poetry
Edgar Allen Poe may or may not have said this.
However, while we’re on the thorny subject of pathogen-induced behavioural changes in the host, it might be worth mentioning a couple of more controversial areas in which it has been proposed.
In the snappily titled paper “Assortative sociality, limited dispersal, infectious disease and the genesis of the global pattern of religion diversity” Fincher and Thornhill argue {{9}} that the wide diversity of religions in the tropics (compared to temperate regions) is driven by infectious disease selecting for three anti-contagion behaviours; in-group assortative sociality; out-group avoidance; and limited dispersal. It’s an interesting idea and I’m pleased I don’t have to test it experimentally. Their argument is that these three behavioural changes select for fractionation, isolation and diversification of the original culture … and hence the evolution of religions.
Conversely, perhaps microorganisms induce religious behaviours (rather than religion per se) that facilitate their transmission. This is exemplified in the entertainingly titled paper “Midichlorians – the biomeme hypothesis: is there a microbial component to religious rituals?” by Panchin et al., (2014). They argue that microbes – and they are really thinking about the gut microbiota here – might be able to influence their hosts (humans) to gather for religious rituals at which both ideas (memes) and infections are more easily transmitted.
Perhaps something to think about when mindlessly spinning out all that summer honey in the next few weeks?
Party, party
I think it’s fair to say that both the papers in the section above have some way to go until they achieve mainstream acceptance … if they ever do.
Furthermore, the general area in which parasites, bacteria and viruses, induce changes in the behaviour of their hosts’ is really in its infancy. We are aware of a lot of behavioural changes, but few are understood at the molecular level {{10}}. As such, we often don’t know whether the association is correlative or causative.
Evolution is certainly a powerful enough selective force to ensure that even extremely subtle benefits to the pathogen may become a genetically-fixed feature of the complex interaction it has with the host.
Respiratory viruses, such as the common cold, Covid-19 and influenza infect millions of people globally and are readily transmitted by direct or indirect contact.
That’s why most of the readers of this post have a face mask nearby and a bottle of hand sanitizer ‘at the ready’. Or should.
Direct transmission benefits the virus as it does not have to survive on a door handle, milk bottle or petrol filling pump.
But direct transmission requires that people meet and are in close contact.
And a paper 10 years ago demonstrated that infection with influenza virus resulted in increased social interactions in the 48 hours post-exposure, compared with the same period pre-exposure {{11}}.
It’s amazing what viruses can do … or might do … or (just look around you) are doing.
{{1}}: That arises by the acquisition of random mutations.
{{2}}: The observable characteristics that result from this interaction.
{{3}}: I’m afraid the paper is behind a paywall and is not Open Access. Disappointing. Complain to the author, not me … ;-)
{{4}}: I know, I know .. it’s dark in the hive, they can’t see anything … but it read better than ‘every bee feeling and smelling’!
{{5}}: So no sneaking off for a cup of tea … this was hard graft.
{{6}}: “Cages” sounds like a barbed-wire enclosure … it was probably a clear yoghurt pot (unused).
{{7}}: Oops, there I go again, smell different!
{{8}}: I. Fries and S. Camazine (2001) Implications of horizontal and vertical pathogen transmission for honey bee epidemiology. Apidologie 32, 199–214.
{{9}}: Proc Biol Sci. 2008 Nov 22; 275(1651): 2587–2594.
{{10}}: But if you want to read more look up some of the stuff on pathogenic fungi.
{{11}}: This is an epidemiology paper and, as a virologist, I think there are a number of problems with it. For there to be a selective advantage the virus would need to be transmissible with these 48 hours. That seems unlikely based upon the known characteristics of ‘flu.



Join the discussion ...