Queen rearing miscellany
Synopsis : Queen cell selection by the beekeeper or the bees – which is more reliable? Nectar collection and comb building by requeening colonies. Three miscellaneous queen rearing topics this week.
Introduction
May to July are the busiest months of the beekeeping season for queen rearing {{1}}. We’re fast approaching the halfway point so I thought I’d write about some related topics, rather than rehash previously covered areas, or pen a magnum opus on just one subject.
This forces me to be a bit less expansive. It means you can skip over less intervening text in the (vain?) hope of finding something of interest … 😉
It also means I should deal with things in less detail.
Alas – I’m writing this introduction after completing the majority of the post – I’ve failed and wrote a lot more than originally intended on the first topic so the miscellany will spill over to next week as well 🙁 .
A loyal listener reader asks …
Fans of Tim Harford’s incomparable More or Less will be familiar with the concept of loyal listeners {{2}}. Since this isn’t a podcast {{3}} listeners is clearly inappropriate.
Unfortunately, I’ve singularly failed to come up with a synonym for loyal starting with an ‘R’, so losing the all-important alliteration with ‘readers’.
Never mind … let’s get back on topic.
One of the pleasures of writing regularly – other than forever playing catch-up with my bloated email inbox {{4}} – is corresponding with beekeepers around the country {{5}}. Sometimes this is in the comments section, but it also involves a considerable volume of email … including many questions or requests for help.
As I’ve previously mentioned, sometimes these exchanges are short and sweet.
Q. What’s the recipe for thin syrup?
A. D’oh! {{6}}
In these instances that might be the only correspondence {{7}} but in other cases there’s a bit of to and fro.
Regular readers {{8}} will recognise some names repeatedly appearing in the comment sections. Many of the questions asked are interesting and some are challenging {{9}}, forcing me to do some thinking and/or reading.
A few allow me to expand further on a topic that I’ve covered, explaining something I either ‘meant to, but ran out of space/time/caffeine’ … or ‘completely forgot’.
And Maccon Keane, a regular reader 😉 from the West of Ireland asked just such a question in the comments to the post last week about beekeeper vs. worker selection of queen cells.
Does beekeeper selection of emergency cells reduce quality?
Here’s the question in full:
Thank you for a really interesting post. My question is this. Using the nucleus method of swarm control by queen removal and induction of the emergency response the beekeeper has to select a queen cell to head the original colony. From these data there is a one in 20 chance (5%) that the chosen cell will not emerge. This is a problem but low risk. However there is a one in two (50%) chance that the beekeeper will select a cell that the bees would have torn down and therefore actively select a lesser quality queen. For an individual colony this may not be particularly significant but over a few generations this negative selective pressure (50%) against the best quality Queens will rapidly lead to a deterioration in stock compared to that which would have happened had the queen been chosen by the bees themselves. Can you think of any way to avoid introducing this systematic negative selection pressure to ensure we let the bees choose the queen because as you title the piece ‘the bees know best’?
This is something I’d thought about, but I’d run out of space to discuss it.
Let’s agree from the outset that the 5% non-emergence rate is an acceptable failure rate. It will be compounded by a small percentage of queens that fail to mate {{10}}.
The beekeeper can’t do very much about either of these.
But what about the beekeeper having a 50:50 chance of selecting a queen cell that the bees would have torn down?
Will this lead to a deterioration of the quality of the bees over time?
It’s an interesting question.
Why do the workers cull about 50% of developing queens?
If you remember, 50% of emergency cells were torn down and these generally contained lighter and smaller queens.
I suggested, or hinted strongly, at three reasons why the bees might favour large queens {{11}} :
- higher fecundity i.e. laying more eggs and/or laying over a longer period
- increased polyandry (and hence colony fitness)
- more likely to survive fights with ‘sister’ queens during polygyny reduction
Fecundity
The researchers addressed this by counting the ovarioles and the volume of the spermatheca. There were no differences between the chosen queens or those that would have been culled. This suggests, though it’s not definitive, that all should have been equally fecund (assuming similar numbers of matings etc.).
You could probably measure this (with sufficient energy, time and money) but it’s not a trivial thing to determine {{12}}. I think the similarity in the number of ovarioles and the capacity of the spermatheca is compelling enough {{13}}.
My assumption is that all, or at least the majority, of queens would be sufficiently fecund to successfully head a colony.
There’s a recent paper on genetic and phenotypic variability of queens that might be useful here, but I’ve not had time to read it properly. If and when I do – if relevant – I’ll update things.
Increased polyandry
I suggested that larger, heavier, queens might fly more strongly, and so spend longer in drone congregation areas or visit more DCAs … and thereby mate with more drones. David Tarpy hints at this in one of the papers cited last week (quoting unpublished results.).
However, I don’t think the work was ever published in a peer reviewed paper as I’ve been unable to find it.
That doesn’t mean it’s wrong {{14}}. Again, it would be a time consuming thing to determine. Queen mating numbers are quite variable so there would have to be a very large number of repeats to get statistically compelling results, but it is doable given sufficient time, money and energy.
Of course, larger/heavier queens might fly less strongly. This hasn’t been tested.
Polygyny reduction
I think this trait is essentially irrelevant in the context of our beekeeping.
By definition we cull all but one developing queen, so the one that is selected should never have to fight another queen. However workers may select for this – perhaps to avoid the risk of two queens fighting and both being damaged/killed – but if they do we can safely ignore it.
Are these ‘lower quality’ queens quantifiably worse for beekeeping?
So, of the three potential differences suggested I’d argue we can rule the last out as being irrelevant (for managed colonies), and we can perhaps safely assume that fecundity will be sufficient (assuming the queen mates with enough drones).
Increased polyandry remains an open question.
So, one possibility is that any queen cell should result in a queen that will be good enough, assuming the queen emerges and mates successfully.
A second possibility is that any differences between the ‘high’ and ‘low’ quality queens – selected from a single colony – are so minor that they have little or no material effect on our beekeeping.
Similar, but not quite the same thing.
It’s worth noting that the only size characteristics (measured) that differed were weight and either thorax length or width. Other dimensions e.g. wing length, were similar.
Is there other evidence to suggest that differences are likely to be minor (with regard to beekeeping)?
In support of this I’d suggest that grafting day-old larvae would not be so universally (successfully) practised if it routinely generated sub-standard queens.
It doesn’t.
When you graft you’re making the selection after less than 24 hours of larval development. The majority of larvae that develop fully, emerge and mate, make perfectly acceptable queens.
But, from a beekeeping perspective, good quality queens are often defined using alternative criteria.
In fact my definition of a good quality queen might well be different from one that the bees would ‘choose’ … or, for that matter, that Maccon would favour.
Selection of good quality stock
And this is where I think selection does have a big influence.
The traits I favour in my bees – steady on the comb, good temper, no following, frugality etc. – vary between my colonies.
I score these traits and preferentially rear queens from the colonies that I consider are my ’best’.
I do this by thirds:
- My ‘worst’ third are always requeened – as soon as is practical – with queens from larvae from my ‘best’ third.
- I similarly requeen my ‘middle’ third with similarly-sourced queens if I have enough spare, but am happy to requeen the ‘middle’ third from the ‘middle’ third (so to speak).
- The ‘worst’ third are never used for queen rearing (or allowed to rear queens from their own larvae). The ‘worst’ third are also discouraged from rearing drones.
If the ‘worst’ third need swarm control I allow them to rear emergency cells, knock them all back a week later – leaving them hopelessly queenless – and then add a frame of eggs/larvae from a better colony.
It’s a guaranteed way to easily improve the quality of your bees.
Which I think pretty much brings me to the end of my answer to Maccon’s question.
In summary … I suspect the difference between queens culled or not by the workers is either irrelevant for our beekeeping, so minor as to be unmeasurable, or swamped by other variables in the mating biology of honey bees (e.g. number of drones available, age of those drones and consequent sperm viability).
Over millennia many factors have resulted in the evolution of the worker selection of developing queens, but over a few ‘honey bee generations’ of managed beekeeping I think we can safely ignore them.
Furthermore, in my opinion, the importance of using a good quality colony as the source of larvae for queen rearing far outweighs the inherent variation in the queens reared from any one colony.
It’s a bit like computing … rubbish in, rubbish out.
Queenless colonies – honey and comb
To close this post on miscellaneous items about queen rearing I thought I’d end with an anecdote and an observation.
The former is supported by little more than my dodgy memory and the latter is backed up by some real science 🙂 .
Foraging efficiency and queenlessness
In Fife the spring honey supers are ready for recovery and extraction. I collected the first batch on Monday and have more to get in a couple of days.
The peak nectar flow seems to have been in the last fortnight of May. Much of it is oil seed rape.
Inevitably, some of the colonies have already had swarm control applied before the peak of the nectar flow. All of my swarm control this year has been using the nucleus method.
At the first sign of swarm preparation (queen cells, either sealed or charged) I make up a nuc with the old queen, destroy any sealed queen cells and leave one charged cell. I return a week later and knock back all but the one selected cell (which is now sealed). The queen subsequently emerges, mates and starts laying.
This means that several colonies have been queenless throughout the peak nectar flow.
All of these colonies have more and/or heavier supers 🙂 .
The queenless colonies seem to have doubled-down on nectar collection and done particularly well this season.
I’ve noticed this before, but it’s really obvious this spring.
My increasingly foggy memory has a dim recollection of beekeepers in the ‘olden days’ removing queens during the nectar flow precisely because they were more productive. I can’t remember when or where I heard/read/imagined this.
Hold on, not so fast
Are they collecting more or just using less because there is no brood to feed? Remember, 8-9 days after applying swarm control, there will be no larvae to feed as all eggs will have developed into sealed brood.
I could do the maths {{15}} but there’s a bunch of assumptions to make about the amount of unsealed brood when the queen was removed etc.
Let’s assume for the sake of argument that a queenless colony stores more nectar because the foragers forage more and because there are fewer hungry mouths to feed in the colony.
Perfect … I’ve got a plan for next season.
I’ll preemptively remove the queens 8-9 days before the main flow and buy 20,000 labels and 6 tons of jars in preparation for a bumper honey crop {{16}}.
But, wait a minute … which are the colonies that usually first start swarm preparations?
That’s right … the strongest colonies.
These are the colonies already filling a double brood box, or overflowing a single brood box.
Perhaps they collect more nectar for the simple reason that there are more foragers?
That’s not the impression I have when I compare the performance of what appear to be equally strong colonies with or without queens. However, ’appear’ is a bit of a loose definition and to be sure I’d need to count frames of brood and the number of foragers.
But it’s an interesting thing to think about {{17}}.
Drawing comb
Another thing I noticed is that queenless colonies provided with foundationless frames continued to draw fresh comb. Clearly they don’t need to have eggs or larvae to occupy the new comb to stimulate comb building.
But the vast majority of the comb drawn was drone comb.
Which, in a roundabout way, led me to this interesting paper:
Smith, M.L. (2018), Queenless honey bees build infrastructure for direct reproduction until their new queen proves her worth. Evolution, 72: 2810-2817.
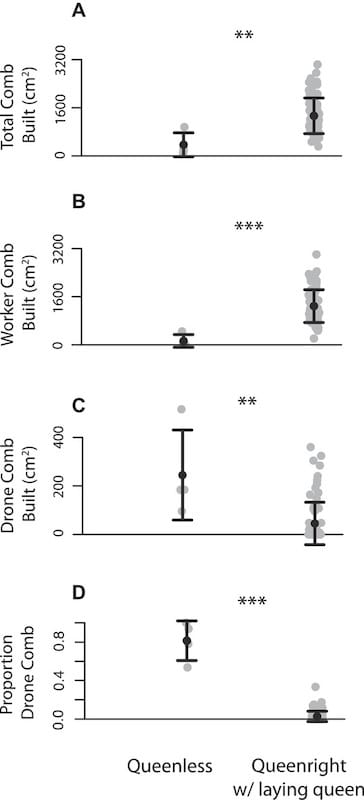
Michael Smith dequeened colonies and investigated whether they built drone or worker comb. The colonies were provided with frames but no foundation (which would otherwise determine the type of comb drawn).
His dequeened colonies built less comb than those with laying queens (A, above), but over 80% of the comb they did build was drone comb (D, above).
Furthermore, they built drone comb even if the colony already contained 25% drawn drone comb (an amount that usually inhibits further drone comb production in a queenright colony).
Finally, he demonstrated that drawing new drone comb only stopped when the colonies contained a new laying queen.
The terminal investment hypothesis
Why should a colony that was queenless or that contained a virgin queen (or for that matter a mated but not laying queen) produce drone comb?
The argument goes something like this.
A colony that is hopelessly queenless can only pass its genes to subsequent generations if it produces laying workers – which lay unfertilised eggs – which consequently develop into drones that mate with virgin queens from other colonies.
The terminal investment hypothesis predicts that the reproductive investment of an individual will change depending upon their reproductive prospects.
Essentially – until there is a laying queen present – the workers pessimistically invest in (i.e. build) drone comb as it offers the only chance of reproductive success should the queen fail to start laying.
Once the queen starts laying they start drawing worker comb again.
As Michael Smith neatly puts it ’When faced with reproductive uncertainty, honey bees may “hope” for the best, but they prepare for the worst’.
And what are the chances of ‘the worst’ happening?
’The worst’ being the failure to replace the queen.
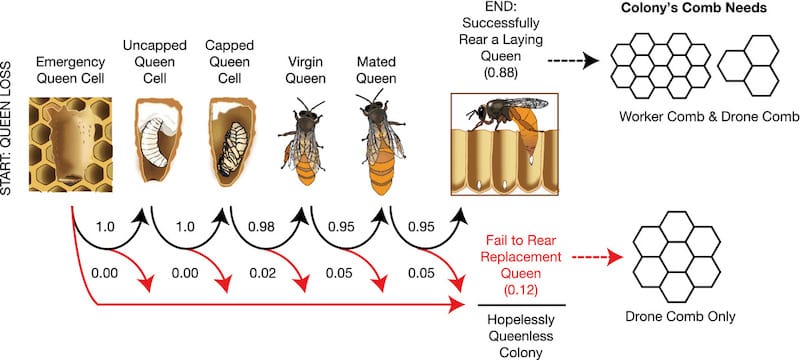
Conveniently Michael Smith also measured the probabilities of successful completion of each of the stages in rearing a replacement queen.
In his studies 98% of queens emerged from the capped cell {{18}}, 95% of virgins returned from mating flights and 95% of those returnees were successfully mated.
0.98 x 0.95 x 0.95 = 0.88 i.e. a queenless colony has an 88% chance of successfully requeening itself, assuming it has eggs/larvae suitable for rearing a new queen.
And the relevance of any of this to practical beekeeping?
- Have confidence during swarm control that the bees will predominantly rear good quality queens (so it doesn’t matter which you choose to keep), if …
- they are good quality bees. And if they’re not then provide them with eggs/larvae from a better colony. You can easily remove deleterious traits and promote good ones. And, if you’ve not got enough (or good enough) colonies to choose from either a) get more 😉 , or b) ’phone a friend’ and scrounge some suitable eggs/larvae.
- Monitor nectar collection by queenright and queenless colonies. Is it different? Many novice beekeepers fret when their colonies are queenless. Maybe at certain times there are benefits 🙂 .
- If you want worker comb, don’t provide queenless colonies with foundationless frames.
- You should assume ~90% of your virgin queens (0.95 x 0.95) will mate successfully and start laying. Always graft a few more larvae than you actually need.
{{1}}: The most fun you can have in a beesuit™.
{{2}}: And, if you’re not, a) perhaps you should be, and b) it’s an easy concept to grasp.
{{3}}: Perhaps it should be? It takes substantially less time to say 3000 words than it does to write 3000 words.
{{4}}: Er … not!
{{5}}: And, increasingly, around the world.
{{6}}: Why ask something that is so easy to look up … you’’ll get the answer faster.
{{7}}: Actually, it almost always is. My emailed reply, however detailed, is rarely acknowledged.
{{8}}: Hmmm … close but no cigar.
{{9}}: Neither description you’ll agree applies to What is the recipe for thin syrup?
{{10}}: Which I’ll return to at the end of this post.
{{11}}: There may well be other reasons as well … and evolution is usually multi-factorial.
{{12}}: I wouldn’t want to apply for the funding to do it.
{{13}}: Particularly since the spermatheca are only filled about 50% during matings – see Tarpy D.R., Keller J.J., Caren J.R., Delaney D.A. Experimentally induced variation in the physical reproductive potential and mating success in honey bee queens. Insectes Sociaux. 2011;58:569–574.
{{14}}: I’ve got a career’s-worth of scientific results that may never be published because they are incomplete, not because they’re wrong (obviously!).
{{15}}: Or I like to think I could … but it’s late and I’m tired.
{{16}}: Which assumes I’ll know when the flow is going to start.
{{17}}: And to understand this more I’m trying to track down Jaycox, E.R. (1970) Honey bee foraging behavior: responses to queens, larvae, and extracts of larvae. Ann. Entomol. Soc. Am. 63: 1689-1694 which I found referenced in an interesting post on Foraging Behaviour by Clarence Collison.
{{18}}: Compared with 95% in the Tarpy studies cited last week.





Join the discussion ...